DIOPTRA seeks to revolutionize colorectal cancer (CRC) screening by offering a non-invasive, accessible, and personalized risk estimation through liquid biopsies. The aim is to identify high-risk cases that require colonoscopy, increase participation rates, and improve early detection.
Why CRC?: Colorectal cancer is one of the top-3 most common cancers, with high mortality rates, metastatic potential, and detectable pre-cancerous states. Current screening participation rates are low, making it crucial to develop better methods.
Objectives:
- Identify a subset of protein biomarkers for early CRC screening and understand their regulatory role through protein network analysis.
- Conduct pilot studies at clinical sites to validate the proposed protocol and refine findings using standardised prospective data.
- Utilize state-of-the-art AI tools to detect significant biomarkers and risk factors related to CRC incidence in an objective and measurable manner.
- Assess the effects of an extended risk factor set on CRC, offering behavioural interventions through a user-friendly mobile tool to impact modifiable pre-malignant states.
- Advocate for change in CRC screening guidelines by providing policy makers with evidence-based data to inform decision-making on potential policy reform.
- Investigate DIOPTRA’s expandability and network with ongoing/future cancer initiatives to empower the newly launched Knowledge Centre on Cancer, enhancing usability and accessibility for early user-friendly screening.
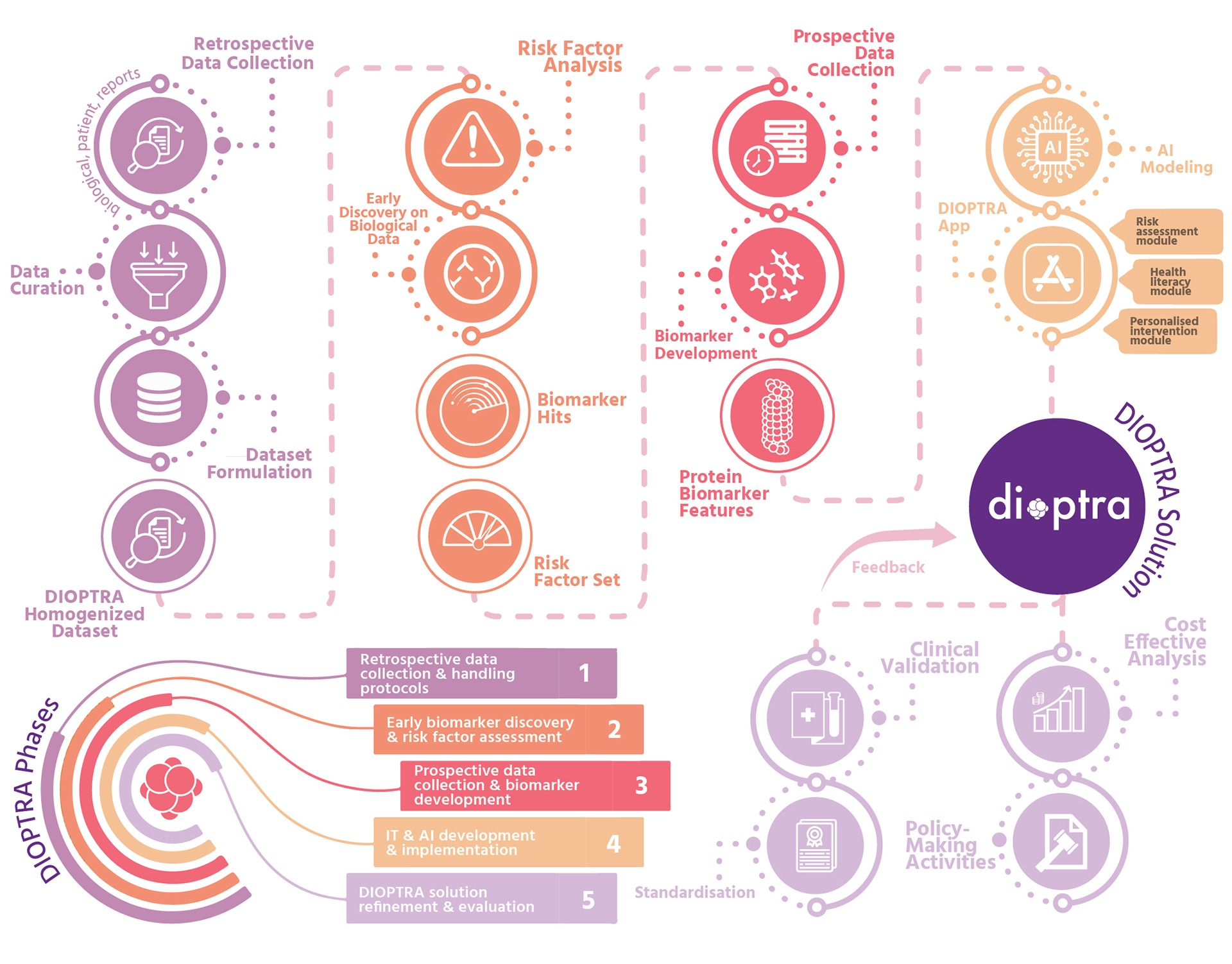
DIOPTRA is organised into 5 distinct phases:
- Phase 1: Retrospective data collection, tissue/liquid biopsy samples, demographic and other relevant data, organise knowledge into standardised format, generate DIOPTRA homogenised dataset.
- Phase 2: Risk factor investigation, initial stage of liquid biopsy biomarker analysis (early discovery), produce holistic CRC risk factor set and set of biomarker ‘hits’.
- Phase 3: First stage of prospective data collection, acquire risk factors information and liquid/tissue biopsy pairs, analyseextended pool of samples for biomarker development, generate protein features for training and evaluating DIOPTRA AI model.
- Phase 4: AI-based analysis for generating discriminative protein biomarkers, develop full stratification model using hybrid approach, incorporate knowledge into DIOPTRA mobile app for personalised behavioural interventions.
- Phase 5: Clinical validation and real-settings feedback, define implementation needs for healthcare upscaling, incorporate full DIOPTRA solution into prospective studies.
